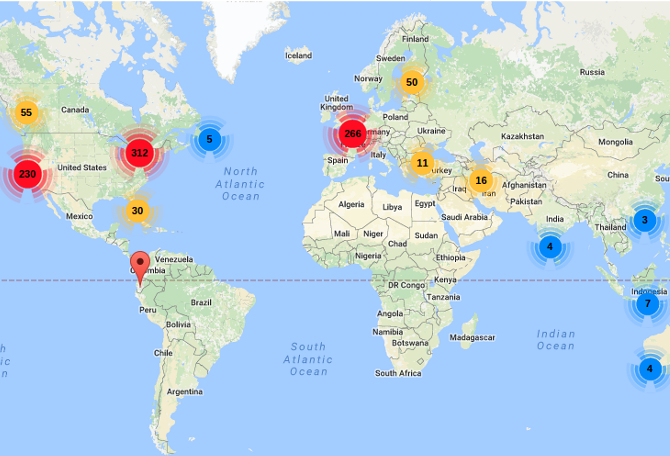
GW Pharma gets FDA panel nod, first cannabis-based drug nearing approval
An advisory panel to the U.S. Food and Drug Administration on Thursday unanimously voted in favor of approving the first cannabis-derived medicine in the country, a childhood epilepsy treatment developed by GW Pharma. The drug, Epidiolex, is derived from cannabidiol (CBD), one of the hundreds of molecules found in the marijuana plant, and an FDA decision is expected by June 27. The syrup contains less than 0.1 percent of tetrahydrocannabinol (THC), the substance that makes people high. The FDA panel found that the drug's benefits outweighed ...
Click
To Read Full Article